

We are developing a battery waste recycling technology based on innovative hydrometallurgy.
Unlike traditional methods using sulphuric acid, our process operates under basic conditions, using an ammonia-based route.
Using the properties of CO2 as a precipitating agent, we selectively extract all the strategic metals present in batteries.
Carbonates
metals
We produce metal salts in the form of carbonates, which have the same properties as conventional metal salts, but without the sulphates, a problematic component both for the environment and for pCAM and CAM manufacturers.
Environnemental performance
This process generates no effluent, making it compact and enabling us to adapt to the quantities of available deposits and to locate as close as possible to waste sources.
Economic performance
By combining environmental performance, our technology enables batteries to be recycled in a virtuous way, while actively supporting the energy transition.
Operation in a basic environment using ammonia, eliminating harmful inputs for reprocessing.
Very little waste is generated, making the plants compact and enabling them to be set up close to the deposits, or even in-situ for gigafactory scraps.
Production of carbonates instead of sulfates, eliminating the environmental and industrial constraints associated with sulfate products.
Our amines are regenerated and reused for continuous metal extraction.
Low input consumption, reducing environmental impact and costs.
The main stages in our process
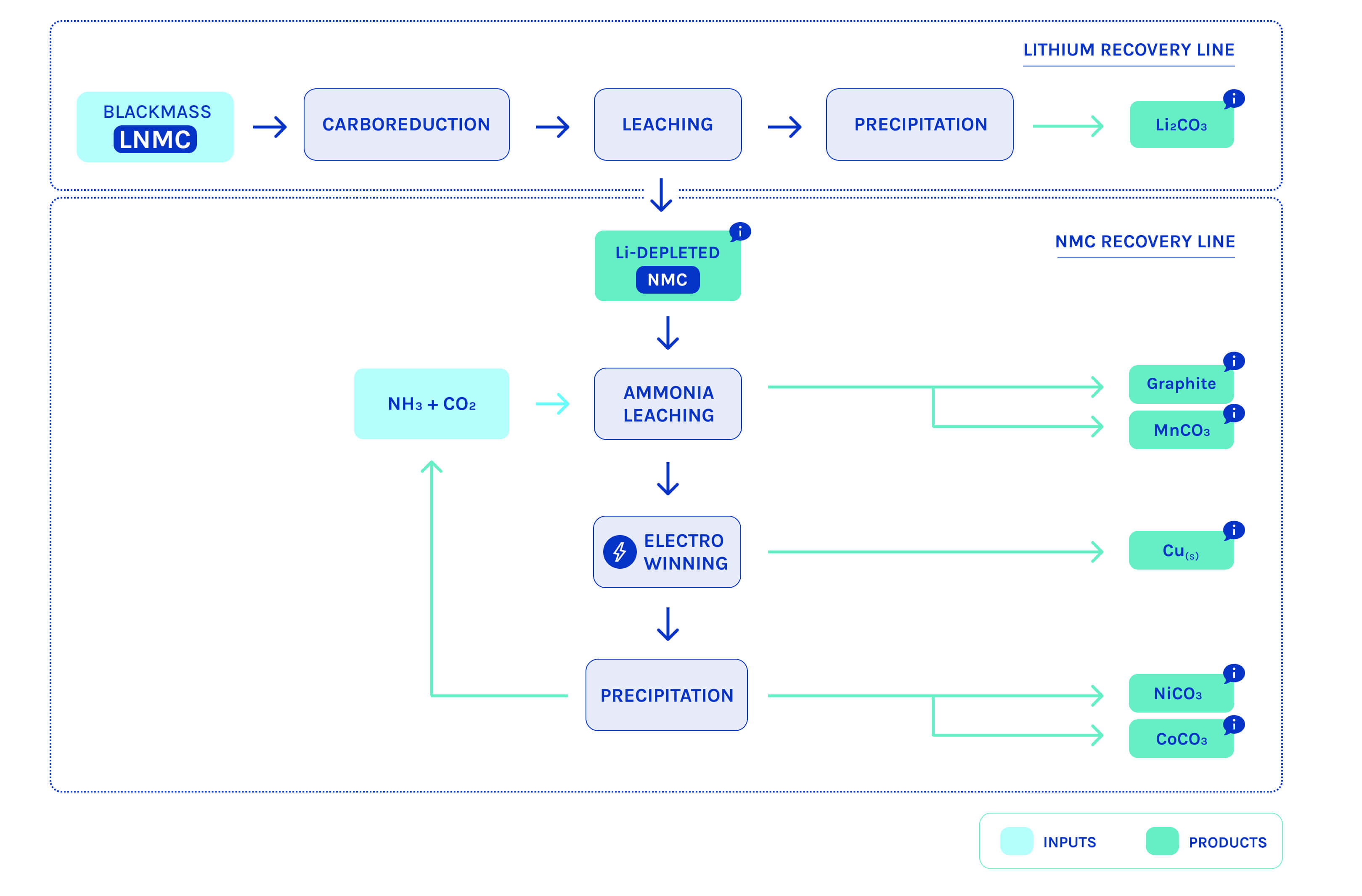






Thanks to our innovative recycling technology, we produce recycled metal salts in the form of carbonates or hydroxides, with optimum purity for reintegration into the battery value chain and for use by industry.
Here are a few examples OF OUR PRODUCTS:

Absolutely! Our process works:
Our tests under pre-industrial conditions show extraction yields in excess of 95% for each metal.
We can extract all the metals contained in Li-ion batteries: lithium, nickel, cobalt, aluminum, copper... We return them in the form of carbonates or hydroxides, which can be used directly to manufacture new batteries.
CO₂ plays a key role in our technology. It is used as a precipitating agent, making it possible to selectively extract each strategic metal (lithium, nickel, cobalt, etc.) without producing complex secondary waste for reprocessing.
Unlike traditional methods based on processes adapted directly from the mining industry, which are energy-intensive and generate waste (pyrometallurgy or acid hydrometallurgy), our :
Mecaware has developed an innovative recycling process based on green, acid-free hydrometallurgy, which uses the properties of CO₂ to selectively extract strategic metals from battery waste. This process operates in a closed loop with optimised environmental and economic impact.
Yes, our technology is modular and adaptable. We can process end-of-life batteries, scraps and other types of metal-containing technological waste.
Our process uses less water and energy than current technologies, making it more sustainable and economically competitive.
Our metal salts can be directly reintegrated into the battery production chain. We work with active material manufacturers (pCAM/CAM producers) to reintroduce them into new cells.
No. Unlike traditional solutions, which require large-scale facilities to be viable, our process is economically viable on a smaller scale, allowing us to adapt to different sources of waste and avoid unnecessary logistical costs.
Our units are compact and flexible, and can be set up close to battery waste deposits or directly in-situ in gigafactories to recycle their production scrap.
We are working to industrialise our technology, setting up pilot and industrial units to demonstrate the performance of our solution on a large scale. We are also continuing to explore new markets, in particular for different battery chemistries (LFP, Na-ion...).
Yes, our process is designed to selectively extract metals, so it could be used for other types of technological waste, such as permanent magnets or electronics in future developments.
Yes, and that's the point! Our metal carbonates and hydroxides meet industry standards and can be used to produce new cathode materials (pCAM/CAM).